We guide engineering teams through the critical early choices that shape performance, quality, and speed. With a focus on risk reduction and process discipline, we enable consistent builds and predictable paths to market.
Design & development
Device projects completed
Combined CDMO experience
Early development sets the tone for everything that follows. Our cross-functional engineers move quickly and collaboratively, blending human creativity with deep technical mastery. The result: devices that take shape with clarity, foresight, and manufacturability already built in. Our skilled teams of engineers and scientists design for usability, function, and performance. All products are built for efficient manufacturing.

We bring your electromechanical concepts to life, designing custom systems that are production-ready and fully integrated.

Forj Medical designs parts that are precise, reliable, and production-ready.

We maintain and enhance your existing devices, addressing reliability, compliance, and new features to enable safer, next-generation products.
Processes are defined, documented, and standardized to ensure designs scale predictably, survive audits, and perform reliably in production. Early integration of engineering and manufacturing improves yield and accelerates the path from prototype to full-scale manufacturing.

We integrate custom automation to simplify assembly operations, improve process capability, and support faster scale-up.

We help teams move complex device designs forward by engineering prototypes and scalable processes at the same time.
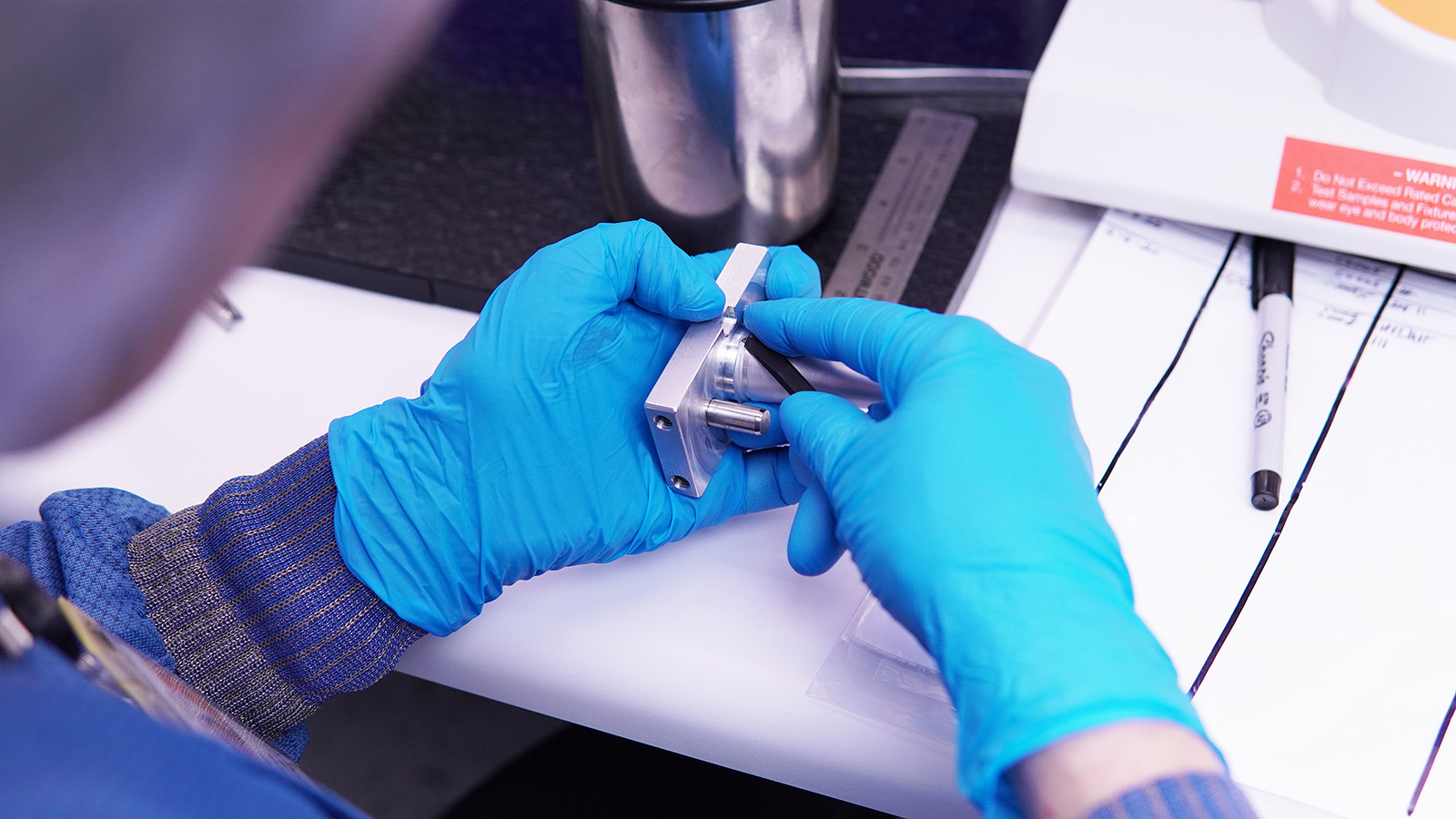
Our structured New Product Introduction (NPI) process accelerates development while minimizing risk. We keep your product moving fast while retaining the highest quality possible, creating a bridge from development to production.
See our application expertise
Uncompromising quality and end-to-end regulatory support from development to production. Our streamlined processes keep projects moving forward, ensuring full regulatory adherence while accelerating timelines.

Our strong and well-established Quality Management System meets all medical device requirements and spans design, development, manufacturing, commercialization, and service of medical devices. Our deep expertise, including REACH, RoHS, conflict, origin materials, and International Harmonized Standards, leads to the delivery of superior medical devices.

Intricon global certification
ISO 13485 certified, FDA registered.
Minnetronix, Inc Certified to:
ISO 13485:2016
FDA Establishment Registration
Compliant to:
ISO 14971
IEC 60601
IEC 62304

We help our partners navigate FDA-related communications, submissions, and meetings and CE submissions, as well as achieving clearances and approvals, ensuring compliance, and mitigating risk.
St. Paul, Minn., and Tacares de Grecia, Alajuela, Costa Rica (March 11, 2026) — Forj Medical, a global contract design...
Advanced engineering collaboration accelerates development of MyBreathPrint™, a revolutionary breath-based cancer detection device and platform that detects lung cancer and...
Forj Medical has announced that Kristin Rodriguez was named Vice President of Human Resources. Rodriguez brings more than 20 years...
Please fill out the form below for unlimited access to all of our case studies.
You now have access to all of our case studies.
Please reach out to us if you’d like to connect with a Forj Medical team member to learn more about any of our case studies or have a specific question to ask.