We combine deep manufacturing expertise with proactive supply chain management to de-risk your program at every stage. With secure sourcing, controlled processes, and responsive operations, we keep your production stable, compliant, and ready to scale.
Insert molded needles
each year
Components and assemblies each year
Manufacturing space across 6 global facilities
Our state-of-the-art manufacturing facilities provide high-volume efficiency through advanced automation, with the flexibility to scale and adapt. We utilize production techniques that reduce waste, elevate quality, and continuously seek efficiencies throughout the process.

We manufacture complete medical device systems, delivering fully integrated, production-ready solutions from concept to commercial scale.

Custom electromagnetic sensor designs to address EM performance and size requirements meet induction, resistance, sensitivity, and localization goals while seamlessly integrating within interventional devices.

Microelectronic assembly and circuit design, incorporating ultra-fine pitch circuits with components down to 0.01005 in., improve performance, reduce cost, and scale efficiently.
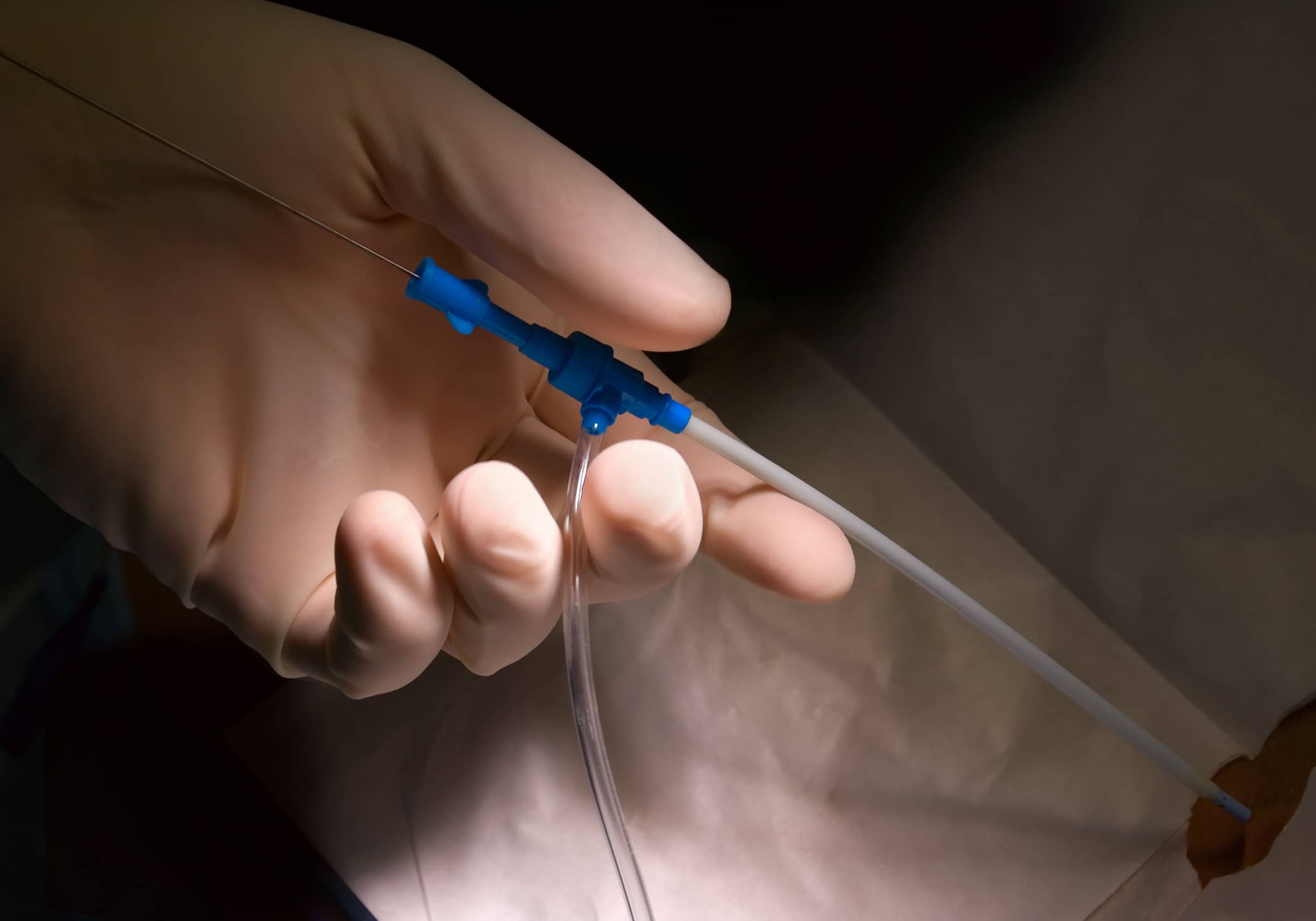
Precision-engineered microcatheters and access sheaths designed for enhanced navigation, trackability, and compatibility with interventional devices improves procedural efficiency, ensures patient safety, and supports next-gen minimally invasive medical treatments.
With design, development, sourcing, and manufacturing operating inside one integrated CDMO ecosystem, teams gain the alignment, transparency, and control needed to manage complexity and deliver high‑performance devices with confidence.

Fully automated high-volume capabilities, best-in-class needle and insert molding capabilities reduce production costs, avoid costly setbacks, improve part quality, and accelerate time to market.

Our in-house PCBA manufacturing line features an end-to-end, fully automated production line and a suite of secondary options that includes quality inspection and functional testing.

Cable assembly capabilities include flexible secondary processing, automated crimping processes, and flexible volumes from small DBRs to full production.

High-precision additive manufacturing for both polymer and metal components accelerates development timelines, reduces material waste, and enables design for intricate medical device components.
Supply chain risk isn’t something we react to, it’s something we plan for. By integrating supply chain strategy from day one, we anticipate obstacles, secure quality materials, and maintain full visibility and control, ensuring reliable, efficient execution without disruption.

From start to finish, we keep your projects moving with precision and care. Our partners choose us for operational excellence, expert guidance, and a level of service that makes working together genuinely seamless.

Global presence, nearshoring efficiency with facilities in the U.S., Asia, and Costa Rica.

We manufacture complete medical devices in ISO-certified cleanrooms that conform to regulatory standards, uphold the highest quality, and deliver precise results.

We design and integrate custom automation into our processes, and the workflow reduces assembly steps, speeds up timelines, and lowers costs for efficient market entry.

Engineered for accuracy, throughput, and scale. Automated inspection, secure logistics, and flexible capacity support efficient production flow.
Please fill out the form below for unlimited access to all of our case studies.
You now have access to all of our case studies.
Please reach out to us if you’d like to connect with a Forj Medical team member to learn more about any of our case studies or have a specific question to ask.